
New research from a team scientists led by Dr. Li Zhang from the Center for High Pressure Science and Technology Advanced Research (HPSTAR) has successfully determined the crystal structure and chemical composition of a deep lower mantle iron-rich phase (H-phase) mineral. The H-phase has the chemical formula Fe₂.₅(OH)₆, with a crystal structure similar to gagarinite. This hexagonal H-phase Fe₂.₅(OH)₆ is currently the known mineral phase with the highest water content under lower mantle cnditions. Its formation under deep lower mantle pressure and temperature conditions is significant for constraining Earth's deep water cycle and explaining the origin of chemical heterogeneity in the lower mantle. The related research was published in the journal Proceedings of the National Academy of Sciences (PNAS) under the title “Gagarinite-type Fe2.5(OH)6 under the deep lower mantle conditions.”
The lower mantle (depth 660–2890 km) is the largest layer by volume in the Earth, connecting the upper mantle and the dynamic core, and plays a key role in Earth's evolution and dynamics. Seismic observations show that the lowermost several hundred kilometers of the lower mantle have a complex structure, with two large low-shear-velocity provinces (LLSVPs) located beneath the Pacific and African plates, and ultralow-velocity zones (ULVZs) only a few tens of kilometers thick at the very base. Further seismic data indicate that superplumes originate from ULVZs at the base of the mantle.
Bridgmanite is the most abundant mineral in the lower mantle, and understanding its physical and chemical properties under deep lower mantle conditions is essential for constraining mantle structure and dynamics. In 2014, Dr. Li Zhang and collaborators first discovered that iron-bearing bridgmanite is unstable under deep lower mantle conditions, decomposing into iron-poor bridgmanite and an iron-rich hexagonal phase (H-phase). This reaction may relate to seismic anomalies in the deep lower mantle.
However, the extreme high-pressure and high-temperature environment of the lower mantle has limited detailed understanding of the H-phase’s crystal chemistry, including its structure, chemical composition, and water storage.
Using the team’s advanced high-pressure polycrystalline diffraction technique, and focused synchrotron X-rays down to<5 μm, they obtained the crystal structure data for the H-phase at 117 GPa and 298 K.
“At pressures of millions of atmospheres, laser heating of mineral samples leads to multiphase coexistence, making diffraction patterns extremely complex. Our high-pressure polycrystalline diffraction technique treats each submicron grain as a single crystal, determining the orientation of each grain, thereby enabling single-crystal analysis under extreme conditions,” explained Dr. Li Zhang.
In this study, laser-heated diamond anvil cells simulated extreme lower mantle conditions—pressures up to 131 GPa and temperatures up to 2200 °C, near the core-mantle boundary. By merging single-crystal diffraction data from three grains, they obtained high-quality crystal structure data for the H-phase.
Crystallographic analysis shows that the H-phase has a structure similar to gagarinite. The mineral is named after Yuri Gagarin, the first astronaut, and normally has the chemical formula NaCa(RE)F₆, where RE represents rare-earth elements (Y, Ce, etc.). Structural refinement and hydrogen content analysis determined that the H-phase has an approximate formula of Fe₂.₅(OH)₆, making it the mineral with the highest known water content under deep lower mantle conditions. The loss of iron from bridgmanite and the formation of Fe₂.₅(OH)₆ may explain chemical heterogeneity and low-velocity anomalies at the base of the lower mantle.
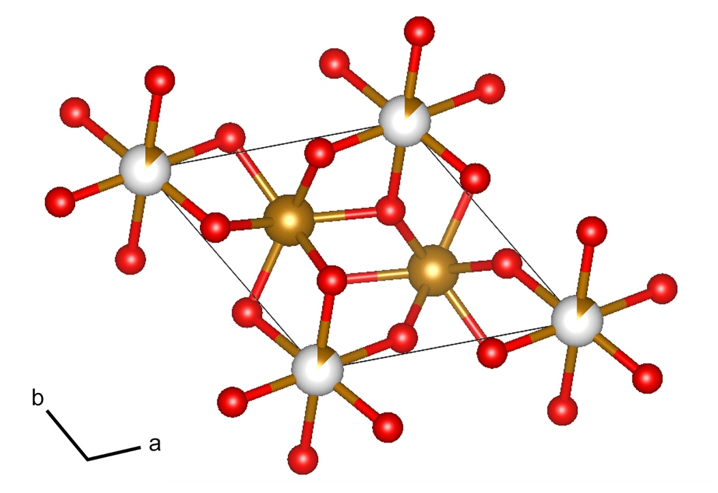
Caption: Crystal structure schematic of gagarinite-type Fe₂.₅(OH)₆ at 117 GPa and 298 K. Gold and red spheres represent iron and oxygen atoms, while white and gold mixed spheres represent partially occupied iron atoms (13.45% occupancy). Hydrogen atoms are not shown. Black lines mark the unit cell.
“Notably, Fe₂.₅(OH)₆ has excellent thermal stability: even at 2200 °C and pressures corresponding to 1000 km above the lower mantle base, it maintains its structure, indicating that Fe₂.₅(OH)₆ is a potential water reservoir phase in the deep lower mantle,” said Dr. Li Zhang.
高压科学研究中心(HPSTAR)张莉团队成功确定了下地幔深部富铁相(H相)矿物的晶体结构与化学组成:H相的化学组成为Fe₂.₅(OH)₆,其晶体结构和加加林石(Gagarinite)的结构类似。具有六方结构的H相Fe₂.₅(OH)₆是目前已知的下地幔条件下含水量最高的矿物相,在深下地幔温压条件下H相的形成对约束地球深部水循环及解释深下地幔化学不均一的起源具有重要意义。相关研究发表于《美国科学院院报》(PNAS)。