
HPNanoPC Group
Research Highlights
Our current research focuses on physical chemical properties of high-entropy and layered materials, and their applications in energy conversion and storage. Here are some research highlights:
1. Electrical Conductivities and Conduction Mechanism of Lithium-Doped High-Entropy Oxides
AI-generated summary: This study explores the electronic and ionic conduction of Li-doped (MgCoNiCuZn)O (Li-HEO) across wide temperature (79–773 K) and pressure (up to 50 GPa) ranges. Li-HEOs are identified as p-type semiconductors where oxygen vacancies create a middle band that influences conductivity. Three temperature-dependent regions are identified: ionization, extrinsic, and intrinsic (above 300 K). Above room temperature, ionic conduction coexists with electronic conduction, making Li-HEO a mixed conductor. While high pressure suppresses ionic conductivity, Li-HEO remains a fast ionic conductor even at 10 GPa. The nonmonotonic effect of pressure on electronic conduction is due to a trade-off between electron repulsion and band approaching. These findings support the use of Li-HEOs in batteries operating under extreme conditions.
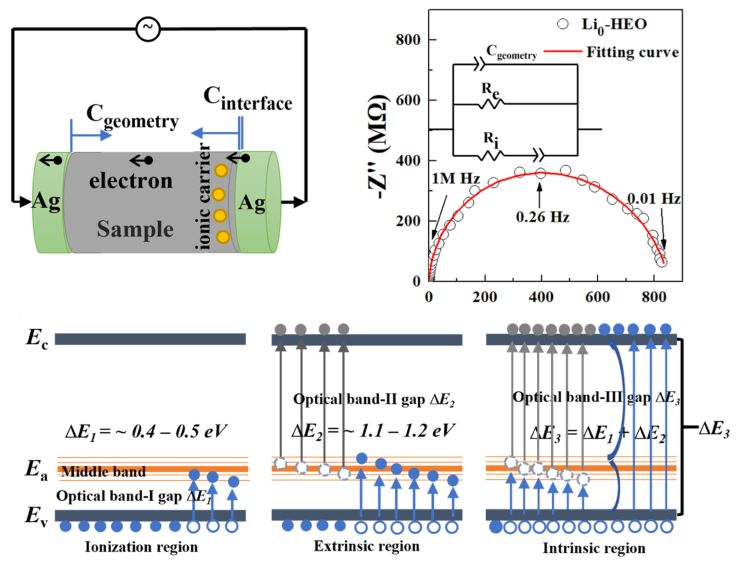
Song, et al., JACS Au 2024, 4 (2), 592–606. https://doi.org/10.1021/jacsau.3c00693.
2. Transition to a Half-Metal Leading to a Drastic Decrease in the Electrical Resistance
AI-generated summary: This research explains the dramatic drop in electrical resistance in (MgCoNiCuZn)O HEO at high pressure without a phase transition. Computational analysis reveals that the HEO transitions from a semiconductor to a half-metal above ~20 GPa. The complex band structure features spin-resolved bandgaps where certain spin bands close under compression, leading to half-metallization. This transition is driven by the emergence of itinerant d–d hopping in Cu and Co 3d states, which bypasses oxygen-mediated paths. Boltzmann transport calculations confirm a shift to a high-mobility regime where electron scattering is inhibited. This work uncovers the HEO's potential as a pressure-sensitive spintronics material and provides an unprecedented view of its spin-related electronic structure.
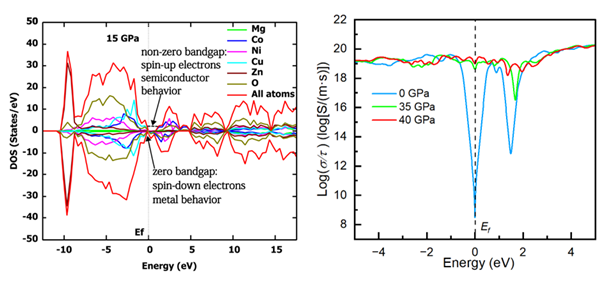
Zhang and Sheng, ACS Omega 2026, 11 (11), 18471–18479. https://doi.org/10.1021/acsomega.6c00843.
3. Metallization and Superconductivity in the van der Waals Compound CuP2Se
AI-generated summary: The study investigates the structural and electrical evolution of the layered van der Waals compound CuP2Se under pressures up to ~60 GPa. It reveals a semiconductor-to-metal transition at approximately 20 GPa and a metal-to-superconductor transition at temperatures of 3.3–5.7 K within the 27.0–61.4 GPa range. Structural changes include isostructural transitions at 10 and 20 GPa, followed by layer sliding and merging into a high-coordination phase between 35 and 40 GPa. Notably, nanosizing occurring under high pressure increases the critical temperature (Tc) during decompression. The superconductivity is linked to enhanced bulk modulus and electron density of states at high pressure, with CuP2Se being paramagnetic and requiring no spin-crossover to achieve a superconductive state.
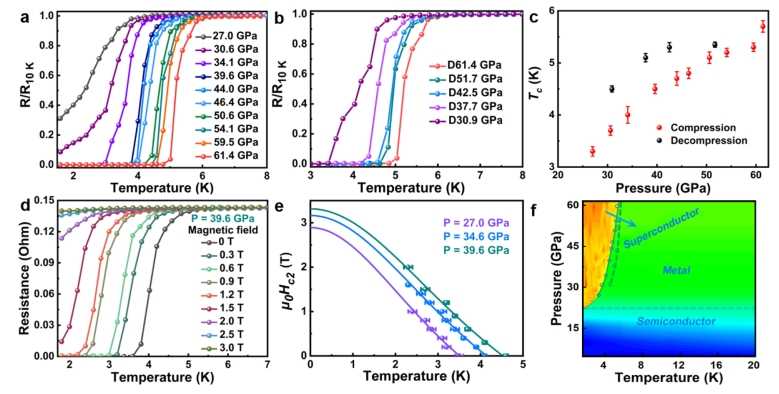
Li, et al., J. Am. Chem. Soc. 2021, 143 (48), 20343–20355. https://doi.org/10.1021/jacs.1c09735.
4. Pressure-Induced Transitions in a High-Entropy van der Waals Compound (MnFeCuCdIn)PSe₃
AI-generated summary: This study examines the high-pressure behavior of a new layered high-entropy phosphoselenide (HEPSe) up to ~60 GPa. The compound undergoes two isostructural changes at ~10 and 20 GPa, followed by a transition to a high-coordination phase at 32–35 GPa. It exhibits a semiconductor-to-metal transition at ~28–30 GPa and a metal-to-superconductor transition (2.5–4.9 K) at pressures between 43 and 58 GPa. The research identifies a linear relationship between the structural transition pressure and cation mixing entropy, and an inverse linear relationship between the superconducting critical temperature (Tc) and entropy. These findings demonstrate that entropy engineering can be used to tune structural stability and superconductivity in complex van der Waals materials.
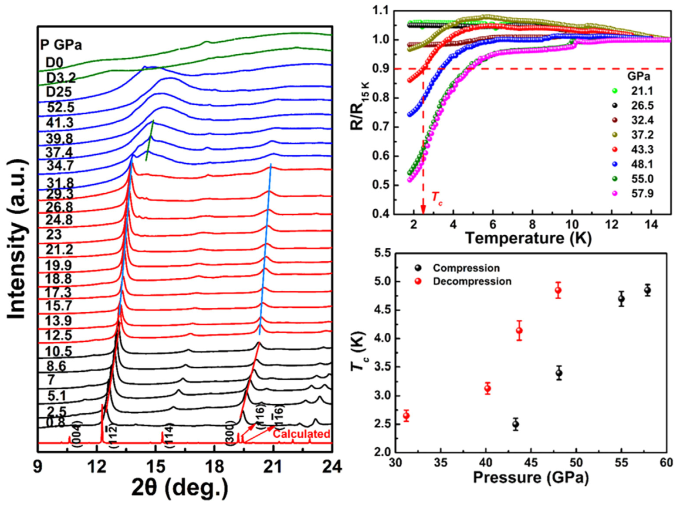
Zhang, et al., Adv. Quantum Technol. 2024, 7 (6), 2300365. https://doi.org/10.1002/qute.202300365.
5. Expediting Oxygen Evolution by Optimizing Cation and Anion Complexity in Electrocatalysts
AI-generated summary: This paper demonstrates that the rate-determining step (RDS) of OER can be intentionally regulated by varying cation and anion complexity in metal phosphorous trichalcogenide (MPT3) catalysts. The synthesized (Ni,Fe)P(S,Se)₃ catalyst exhibited exceptionally high activity, characterized by an ultra-low Tafel slope of 34 mV/dec. This performance is attributed to a shift in the RDS from the formation of M–O* to M–OOH* species. The authors proposed a complexity index based on pairwise metal-S/Se bond contributions that scales linearly with catalytic activity descriptors. This index allows for the prediction of OER activity based simply on chemical composition rather than complex computations. The study confirms that the reacting interface, rather than surface oxyhydroxides, provides the active sites.
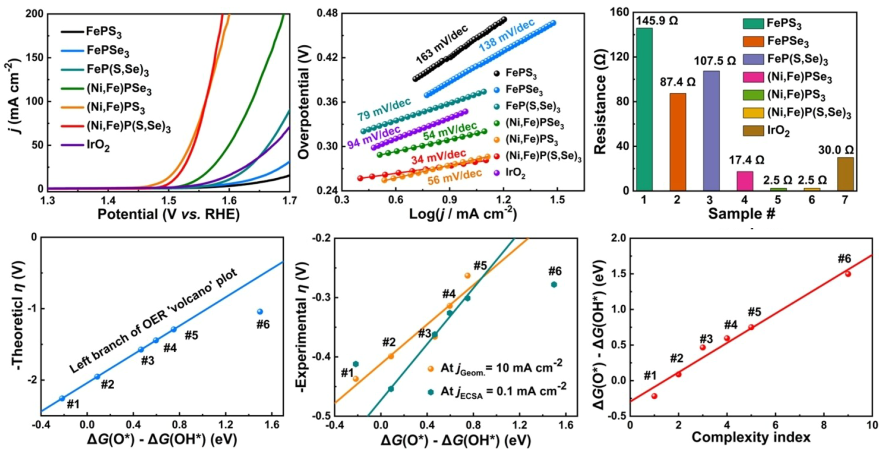
Li, et al., Angew. Chem. Int. Ed 2023, 62 (9), e202214570. https://doi.org/10.1002/anie.202214570.